Method overview
Intermittent Hypoxic Treatment – What is it?
Intermittent/Interval Hypoxic Training or Intermittent/Interval Hypoxic Treatment (IHT) – a commonly employed designation of a treatment method using intermittent hypoxic exposure as the main therapeutic factor.
An IHT session consists of an interval of several minutes (3–10 min each) breathing hypoxic (low oxygen) air, alternated with similar intervals breathing ambient (normoxic) or hyperoxic air (Hypoxia – Hyperoxia mode – IHHT).
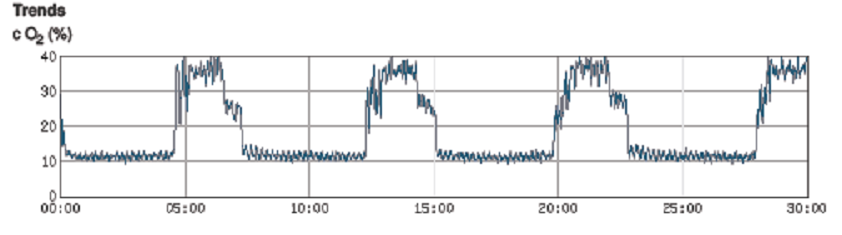
The procedure may be repeated several times in variable-length sessions per day, depending on a physician’s prescription or a manufacturer’s protocol (Navarrete-Opaz et al.,2014). Standard practice is for the patient to remain stationary while breathing hypoxic air via a hand-held mask. The therapy is delivered using a hypoxicator during the day time, allowing the dosage to be monitored. https://en.wikipedia.org/wiki/Intermittent_hypoxic_training
Unlike climbing a mountain, where the body adapts to gradually lower levels of oxygen, IHT deprives the body of oxygen in short, sharp bursts. During treatment, oxygen saturation levels in the blood, heart rate, and blood pressure are closely monitored. This enables oxygen levels to be reduced safely.
The main idea of the interval hypoxic treatment method is repeated reduction of blood oxygen to the individual hypoxia adaptation level intermingled with recovery intervals.
A reduction in oxygen level stimulates activity of protective proteins (e.g. antioxidant enzymes, heat shock proteins, Fe-regulating proteins, repair enzymes, growth factors,peroxiredoxins, etc. by starting a cascade of reactions that leads to hypoxia adaptation and new defensive mechanisms formation which produce positive physiological changes and outcomes in clinical practice [Bernardi et al.].
When intermittent hypoxia can be useful
Therapeutic potential of HIF signaling pathway: “a matter of dose”
Only the controlled and dosed exposure to normobaric hypoxia may be used as a therapeutic method.
The efficacy of IH is dependent upon five key variables (Törpel et al.2019):
- the intensity of hypoxia
- the duration of hypoxia
- the number of cycles
- the pattern of IH presentation (e.g., consecutive days vs. alternating days)
- the total protocol duration
Alexander Törpel et al. (European Journal of Applied Physiology, 2019):
“While the external intensity of hypoxia determines the type of the environmental condition (e.g., the fraction of inspired oxygen: FiO2), the internal intensity of hypoxia is characterized by the amount of oxygen deficit in the organism (e.g., the oxygen saturation of the blood: SpO2, measured by a pulse oximeter). Here, the intensity of internal hypoxia depends among others on the intensity of external hypoxia, some physiological processes (e.g., lung diffusion capacity), the chemoreceptor sensitivity (García-Río et al. 2007) and the compensatory response of the organism (e.g., the cardiorespiratory system; Bärtsch and Gibbs 2007). With regard to the latter three factors, inter-individual differences and age-specific changes can be determined (García-Río et al. 2007; Lhuissier et al. 2012; Stam et al. 1994). Consequently, there are inter-individual differences of the internal intensity of hypoxia during the administration of an external hypoxia which may result in a different severity of the hypoxic stimulus for the organism (variations by up to 10% or more of the SpO2, see Chacaroun et al. 2017; Burtscher et al. 2004; Harshman et al. 2015).
SpO2 of about 80% corresponds to the average internal intensity of hypoxia during an external hypoxia with a FiO2 of 17–10% (see Burtscher et al. 2004; Chacaroun et al.2017).
Modest hypoxia (10-16% inspired O2) and low cycle numbers (3-15 episodes per day) most often lead to beneficial effects without developing any pathological changes, whereas severe hypoxia (2-8% inspired O2) and more hypoxic episodes per day (48-2400 episodes/day) elicit progressively greater damage. Accumulating evidence suggests that “low dose” IH (modest hypoxia, few episodes) may be a simple, safe and effective treatment with considerable therapeutic potential for multiple clinical disorders. (A. Navarrete-Opazo and G. S. Mitchell, Department of Comparative Biosciences, University of Wisconsin-Madison, Madison, Wisconsin, 2014).
Some facts about Hypoxia Training
- The Hypoxia Therapy method is very natural, as it utilizes unique genetically installed human body phenomena: an organism rapidly compensates disruptions at all levels caused by decreases in oxygen intake, adapting to its new environment. So-called ‘super-recovery’ follows total recovery of a disrupted function so that body resistance to a specific factor improves.
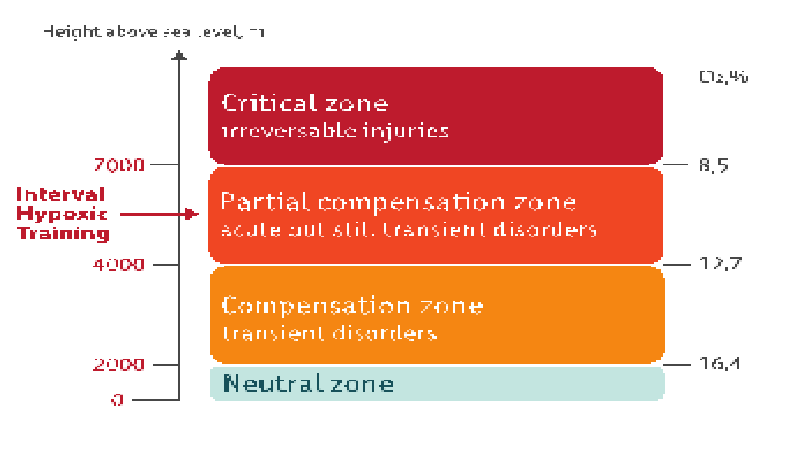
- Prenatal body evolution takes place in a low PO2 environment similar to an altitude of 5,000-6,000m above sea level (asl). Furthermore, PO2 values in the womb and fetal tissues change cyclically.
- Ability of the fetus to super-recover in response to decreased PO2 is an aspect of specific fetus «training» and forms its ability to resist external factors following parturition.
- Intermittent Hypoxia Therapy is a convenient way to tune the oxygen transport system and increase the power of the oxidative process. It relates to the fact that the oxygen the transport system, including breathing, cardiovascular systems, blood, multilayered regulators and cell breath systems dominate in terms of responsibility for adaptive changes.
- These effects are realized also during continuous intensive physical exercises of aerobic nature of longer than 30 minutes. Hypoxic training is easy to perform, which along with the absence of limitations is another benefit for patients, especially those overweight or with metabolic syndrome.
- Highland communities typically enjoy a 15–20 years longer lifespan. Median lifetime of Hunz valley inhabitants, in the Karakorum mountains, at 2,500m asl, is 120 years.
Physiological effects
Mechanism of short-term adaptation to hypoxia
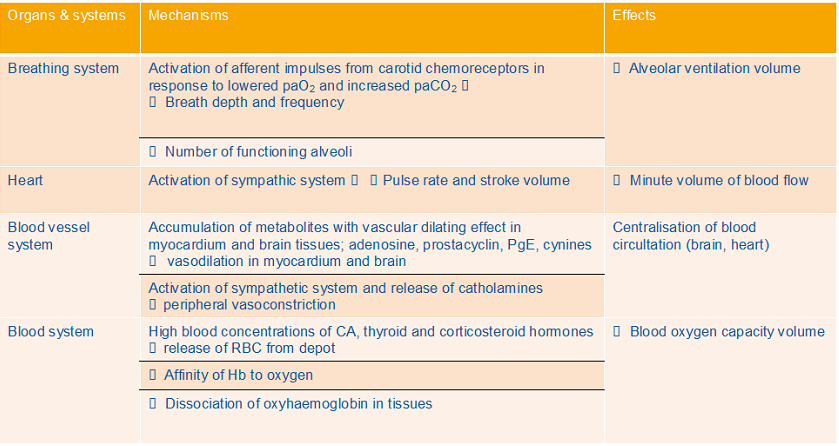
Compensatory mechanism of adaptive responses to interval hypoxia
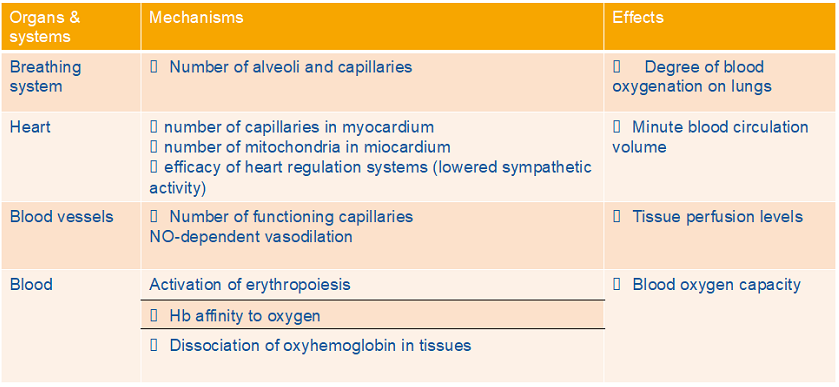
IHT/IHHT Applications
The intermittent exposure to hypoxia during IHT stimulates epigenetic changes and increased expression of genes that allow adaptation to altitude. The end result of hypoxia adaptation includes improved oxygen, better circulation, improved mitochondrial function, increased tolerance to various stressors and even toxic chemicals, increased antioxidant production and reduced inflammation (Kayser and Verges 2013, Verges, Chacaroun et al. 2015). These adaptive changes can enhance physical and mental capacity, so that the body is better able to cope with a range of stressors and repair and heal cells tissues and organs. (Serebrovskaya, Nikolsky et al. 2011) .
IHT increases endothelial production of nitric oxide (NO) while also preventing NO overproduction in the brain and other tissues. The end result is neuroprotection of various pathologies, reduced blood pressure (Lyamina, Lyamina et al. 2011), and a reduction of oxidative stress (Malyshev, Bakhtinia et al. 2001, Manukhina, Downey et al. 2016).
IHT/IHHT effects:
- Increased tolerance to physical load
- Increased VO2 max – maximum rate of oxygen usage
- Enhanced power output and speed
- Increased exercise-until-exhaustion (ETE) time
- Improved quality of life
- Increased activities of daily living
- Reduced Depression symptoms
- Improved the quality of sleep
- Decreased blood pressure and heart rate
- Cognitive performance improvement
- Reduced possibility of negative effects of the acute hypoxia/ ischaemia – increased myocardium and brain resistance to acute ischaemia (preconditioning effect)
Application
In patients
- Rehabilitation (cardiovascular/pulmonary /spinal cord inquires)
- Dementia/ Alzheimer’s disease
- Metabolic Disorders (Diabetes Mellitus, Prediabetes,Obesity)
- Sport Medicine
- Healthy
- Preventive & Anti-aging Medicine
- Sport& Fitness
- Wellbeing
- Burnout and exhaustion
- Sleep disturbances